Pfizers vaccine is administered as two 30-microgram doses given 21 days apart. Between 21 December 2020 and 18 December 2021 some 75 million doses of Moderna vaccine were administered in the US with 19 anaphylactic reactions recorded 25 casesmillion doses administered.
Everything You Need To Know About The Moderna Covid 19 Vaccine Chemist Druggist
The FDA expanded the emergency use authorization of the Pfizer-BioNTech COVID-19 Vaccine to include adolescents 12 through 15.
Moderna vaccine review date. Compliance with the full schedule is recommended and the same product should be. The UK has bought 7m doses of the Moderna vaccine. SAGE recommends the use of the Moderna mRNA-1273 vaccine at a schedule of two doses 100 µg 05 ml each 28 days apart.
The vaccine was also highly effective in clinical trials at preventing COVID-19 among people of diverse. Written by Minseo Jeong on May 6 2021 Fact checked by Harriet Pike PhD. The Moderna COVID-19 vaccine is a two-dose vaccine to prevent.
April 5 2021 414 AM 4 min read. Mar 11 2021 114 PM Ian Haydon was one of the first people to try out Modernas COVID-19 vaccine as part of the companys phase-one trial with. The following have been updated to reference the Interim Clinical Considerations for the Use of COVID-19 Vaccines Currently Authorized in the United States from May 14 2021.
As of 9am on January 8 2021 more than 66 million people in the United States have received their first dose of. European Medicines Agency EMA the blocs medicines watchdog said on Thursday it had brought forward the date for the assessment of a marketing authorisation application for Modernas coronavirus vaccine by nearly a week to 6 January. The Moderna Vaccine Preparation and Administration Summary.
The Moderna COVID-19 Vaccine is a vaccine and may prevent you from getting COVID-19 the virus that causes fever or chills. Notably none of these allergic reactions were fatal. EMA brings forward Modernas vaccine review date.
Modernas vaccine is administered as two 100-microgram doses given 28 days apart. To date there have been no serious or long-term side effects associated with either the Pfizer or the Moderna vaccine. Dosage and timing.
Final results from the trials of Modernas vaccine against Covid-19 confirm it. If necessary the interval between the doses may be extended to 42 days. Yes the coronavirus vaccine is safe.
The Pfizer and Moderna vaccines are administered in two doses. However according to the Pew Research Center as of December 3 2020 39 of US adults say they would probably not get a coronavirus vaccine. As such the quoted 2023 date for the completion of the Moderna vaccine in the facebook post is incorrect.
17-Jun-2021 Data Lock Date. The Moderna COVID19 vaccine codenamed mRNA-1273 and sold under the brand name Spikevax is a COVID-19 vaccine developed by Moderna the United States National Institute of Allergy and Infectious Diseases NIAID and the Biomedical Advanced Research and Development Authority BARDA. The Moderna COVID-19 Vaccine Standing Orders.
17-Jun-2021 Page 1 COVID-19 Moderna vaccine analysis print Report Run Date. 16-Jun-2021 183009 All UK spontaneous reports received between 91220 and 160621 for Moderna vaccine. How Well the Vaccine Works.
Based on evidence from clinical trials in people aged 18 years and older the Moderna vaccine was 941 effective at preventing laboratory-confirmed COVID-19 infection in people who received two doses and had no evidence of being previously infected. It will be the second such meeting in 8 days of the FDAs Vaccines and Related Biological Products Advisory Committee VRBPAC which will review an. The trial completion dates are set in the future to allow for long term follow up however analyses of the data from phase three trials have been published in peer-reviewed studies in medical journals.
It is used in people aged 18 years and older to provide protection against infection by the.
Covid Vaccine Moderna Says Its Shot Is 90 Effective 6 Months After Second Dose

Pfizer Seeks Full Fda Approval For Covid 19 Vaccine Coronavirus Updates Npr

5 Ri Hospitals Get Shipments Of Moderna Vaccine After Subcommittee Grants Approval Wpri Com

Coronavirus Vaccine Pfizer Moderna And How Many Vaccine Doses Are Coming In 2020 Cnet
/cloudfront-us-east-2.images.arcpublishing.com/reuters/GI3BFBIIVRJEPKM6GVUJJZNU3I.jpg)
European Medicines Regulator Brings Forward Moderna S Vaccine Review Date Reuters

Cutaneous Reactions Reported After Moderna And Pfizer Covid 19 Vaccination A Registry Based Study Of 414 Cases Journal Of The American Academy Of Dermatology
Coronavirus Vaccine What Is The Current Status Of Moderna Covid 19 Vaccine And Everything You Should Know About It The Times Of India
Covid 19 Vaccine Efficacy Summary Institute For Health Metrics And Evaluation
Covid 19 Vaccine Information Stamford Ct

Germany Restricts Use Of Astrazeneca Vaccine To Over 60s In Most Cases News Dw 30 03 2021
Pfizer Covid Vaccine Is 95 Effective Plans To Submit To Fda In Days
Fda Staff Recommends Watching For Bell S Palsy In Moderna And Pfizer Vaccine Recipients

How The Fda Authorized Moderna Covid 19 Vaccine Compares To Pfizer S Science News
Covid Vaccine U S Plans To Ship 6 Million Moderna Doses Once Fda Gives Ok
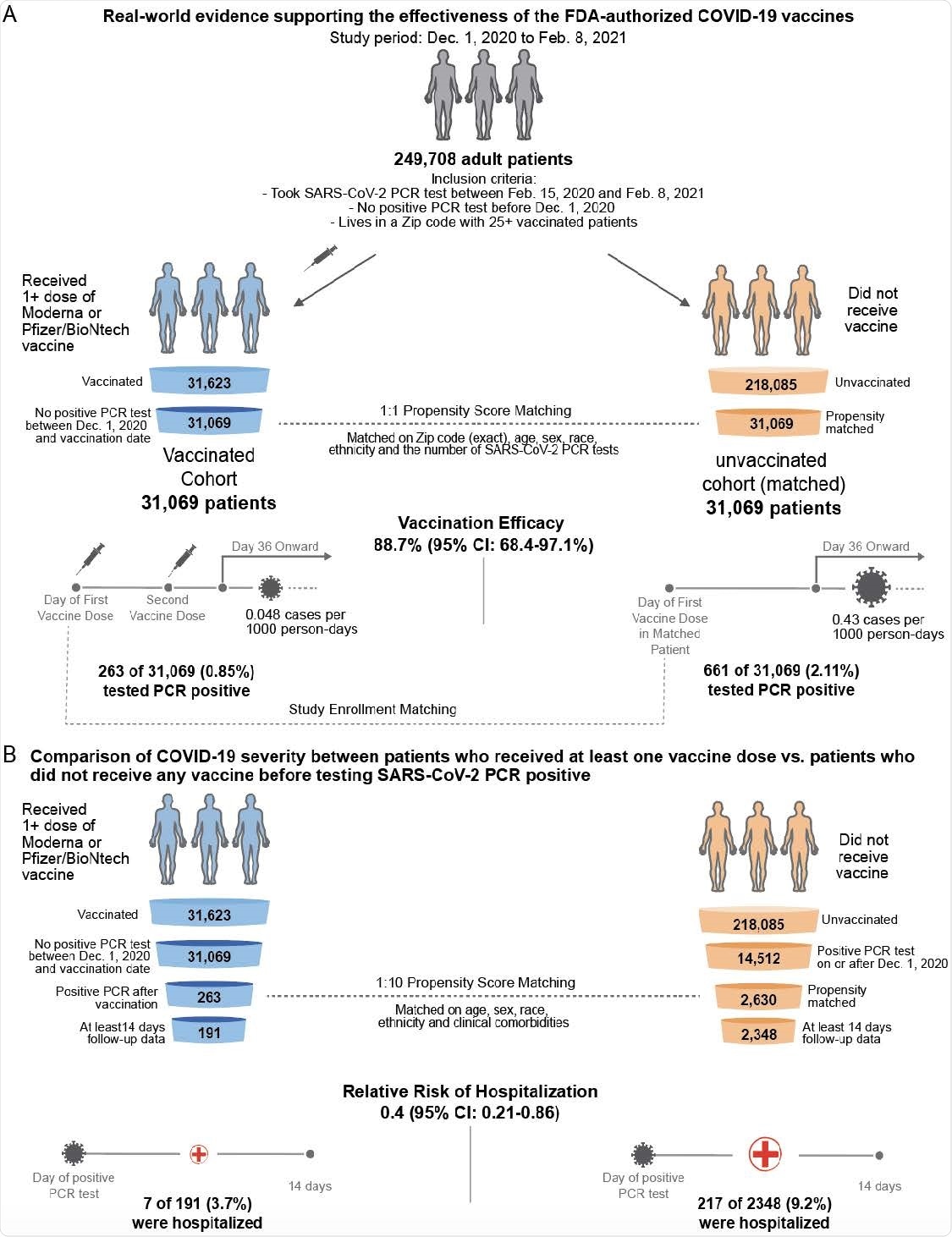
Study Shows Real World Effectiveness Of Moderna And Pfizer Biontech Vaccines

Incidental Illnesses Threaten To Give Covid 19 Vaccines A Bad Rap
Covid Vaccine Fda Staff Endorses Emergency Use For Moderna In Key Step Toward Approval
What You Need To Know About Astrazeneca S Covid 19 Vaccine Science In Depth Reporting On Science And Technology Dw 18 03 2021


